
OrthogenRx CEO & Founder Michael Daley talks about the Company’s new 3-injection HA product, TriVisc...

Elise Wolf, SmartTRAK's Senior Analyst, Computer Assisted Surgery, will be representing SmartTRAK at CAO...

Frederique Buntoum (aka Freddy), SmartTRAK's Senior Analyst, Regenerative Medicine, overseeing the Cell ...
.png)
SmartTRAK examines what the combined company will look like in terms of revenue, product portfolios, fin...

Dr. Ronald Baxter of Baylor University and Spectral MD, discusses the Spectral MD DeepView Wound Imaging...

As surgeries get pushed to later in the year, growth against easier comps does not signal market strengt...

SmartTRAK interviews Arthrosurface CEO Steve Ek about the Company’s newest shoulder system, the OVOMotio...

The SmartTRAK analyst team is expanding and we are looking for an experienced and motivated individual w...

Intraoperative neuromonitoring could be a game changer in the field of SCS by elucidating the MOA for cu...
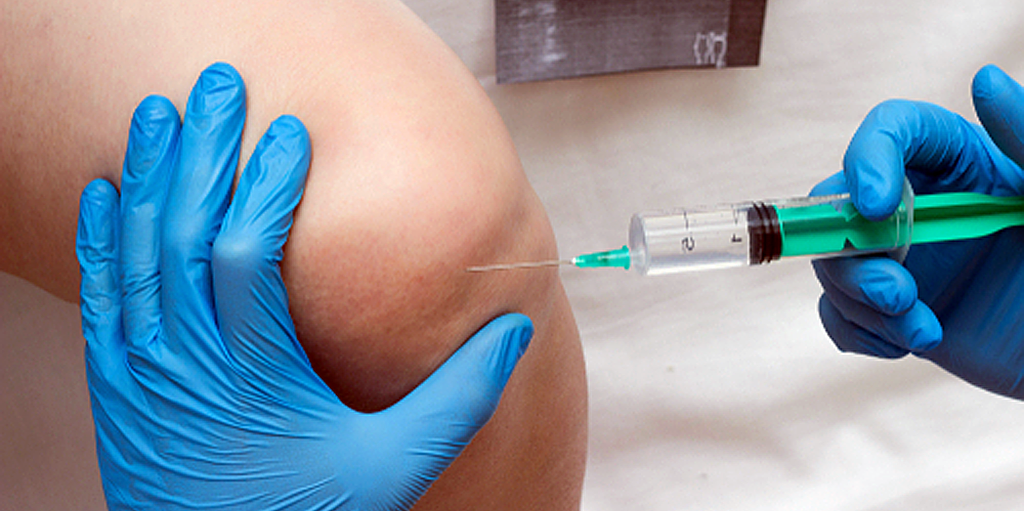
Revenues down again, competitive pricing pressures remain, market is under FDA scrutiny After a sluggish...